COVID19 Update: May 5, 2020
COVID-19 update for Tuesday, May 5 as of 3:00 p.m.
Pennsylvania Update
Governor Wolf: Reopening Pennsylvania
Governor Wolf has provided guidance to businesses as 24 counties move to the “yellow phase” of reopening beginning this Friday, May 8. The guidance details procedures businesses must follow to conduct in-person operations. See the administration’s announcement of the guidance here and go here for the guidance itself.
Department of Human Services
 During its April 29 COVID-19 webinar, DHS officials explained that the department has accelerated certain capitation payments to Medicaid MCOs. This action will enable the advance in appendix 14 and appendix 17 payments described previously, which are expected to be distributed to hospitals on June 4.
During its April 29 COVID-19 webinar, DHS officials explained that the department has accelerated certain capitation payments to Medicaid MCOs. This action will enable the advance in appendix 14 and appendix 17 payments described previously, which are expected to be distributed to hospitals on June 4.- During that same webinar, DHS officials also discussed a recent change in prior authorization requirements for long-term acute hospital admissions.
- DHS’s Office of Medical Assistance Programs has updated its FAQ about coverage of COVID-19 testing and related services for participants in the state’s CHIP program.
Department of Health
The Department of Health has published interim guidance on conditions and circumstances for discontinuing non-health care isolation for persons with COVID-19.
The department has written to the leaders of hospitals, health care facilities, and laboratories asking them to complete a survey that will be used by the state to determine Pennsylvania’s current capacity for administering COVID-19 tests. See the letter here and the survey here.
Department of Health Daily Briefing
 After reconciling death count data over the past two weeks, including data from Philadelphia, the state has added 554 new deaths to its total count. This raises the total death count nearly 23 percent over yesterday’s announced total.
After reconciling death count data over the past two weeks, including data from Philadelphia, the state has added 554 new deaths to its total count. This raises the total death count nearly 23 percent over yesterday’s announced total.- Among the nearly 51,000 Pennsylvanians who have been diagnosed with COVID-19 are 3200 health care workers, more than 9600 residents of long-term-care facilities, and more than 2000 workers in food processing facilities.
- 2583 Pennsylvanians are currently hospitalized with COVID-19 and 542 of them are on ventilators.
- 46 percent of the state’s acute-care hospital beds and 40 percent of its ICU vents are unoccupied and nearly 75 percent of its ventilators are idle.
- Secretary Levine told a legislative committee that the state would not provide personal protective equipment to hospitals that have resumed performing non-urgent procedures because one of the conditions for resuming such procedures is having an adequate supply of such materials.
Federal Update
Centers for Medicare & Medicaid Services
- CMS has updated its FAQ for state Medicaid and CHIP programs. Among the issues the FAQ addresses are emergency preparedness and response, eligibility and enrollment flexibilities, benefit flexibilities, cost-sharing flexibilities, financing flexibilities, managed care flexibilities, information technology and data reporting. A separate document includes only the new questions.
Centers for Disease Control and Prevention
 The CDC has posted guidance for nasal (anterior nasal) specimen collection for COVID-19 diagnostic testing.
The CDC has posted guidance for nasal (anterior nasal) specimen collection for COVID-19 diagnostic testing.- The CDC has updated its interim guidance for collecting, handling, and testing clinical specimens from persons with COVID-19.
- The CDC has updated its strategies for optimizing supplies of personal protective equipment.
- The CDC has posted general guidance for handling bulk-packaged sterile swabs properly for upper respiratory sample collection during the COVID-19 crisis.
- The CDC has posted guidance for proposed use of point-of-care testing platforms for COVID-19.
Food and Drug Administration
- The FDA has updated its policy on the review and approval of diagnostic and antibody tests for COVID-19. In a commentary titled “Insight Into FDA’s Revised Policy on Antibody Tests for COVID-19,” FDA officials explain the evolution of the agency’s approach to issuing emergency use authorizations for antibody tests. The new policy itself can be found here.
- The FDA has issued emergency use authorizations (EUA) for two specific commercial diagnostic tests for COVID-19. See them here and here.
Medicaid Health Plans of America
- Medicaid Health Plans of America, a trade association that represents more than 90 Medicaid managed care plans serving Medicaid beneficiaries in 37 states, has written to leaders of Congress to ask that they increase the federal medical assistance percentage (FMAP), the rate at which the federal government matches state Medicaid expenditures, by 12 percent retroactive to January 1, 2020 and through September 30, 2021. The group also asked Congress to delay or cancel CMS’s Medicaid fiscal accountability regulation (MFAR).
Resources to Consult
Pennsylvania Department of Human Services
Pennsylvania Department of Health
Centers for Disease Control and Prevention
(To receive this daily update directly, sign up for our mailing list at info@pasafetynet.org.)
 With 825 new cases yesterday, Pennsylvania has surpassed 50,000 people diagnosed with COVID-19. 2500 of them have passed away.
With 825 new cases yesterday, Pennsylvania has surpassed 50,000 people diagnosed with COVID-19. 2500 of them have passed away. Department of Health and Human Services
Department of Health and Human Services The Department of Labor has issued additional guidance about 100 percent federal reimbursement of certain state short-term compensation payments and other changes in short-term compensation programs. The guidance seeks to address “…how states can take advantage of this program as they look to re-open their businesses.” For further information see the department’s
The Department of Labor has issued additional guidance about 100 percent federal reimbursement of certain state short-term compensation payments and other changes in short-term compensation programs. The guidance seeks to address “…how states can take advantage of this program as they look to re-open their businesses.” For further information see the department’s 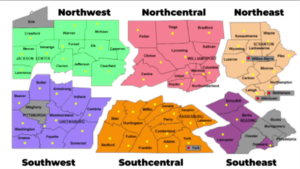 Governor Wolf on Friday announced the reopening of 24 Pennsylvania counties in the northwestern and north-central parts of the state, moving them from red to yellow in his three-color plan for reopening the state. The governor outlined the criteria used to reach these decisions and the provisions for contact tracing and additional testing that make reopening possible. The reopening calls for reduced use but not elimination of social distancing and includes guidance on work, congregate, and social activities that are now permitted as well as those that remain restricted or prohibited. On Monday the administration will release guidance for businesses permitted to reopen in the selected counties.
Governor Wolf on Friday announced the reopening of 24 Pennsylvania counties in the northwestern and north-central parts of the state, moving them from red to yellow in his three-color plan for reopening the state. The governor outlined the criteria used to reach these decisions and the provisions for contact tracing and additional testing that make reopening possible. The reopening calls for reduced use but not elimination of social distancing and includes guidance on work, congregate, and social activities that are now permitted as well as those that remain restricted or prohibited. On Monday the administration will release guidance for businesses permitted to reopen in the selected counties. Yesterday CMS released a second interim final rule with comment period announcing a new round of regulatory waivers and rule changes to provide additional flexibility to the health care system as the COVID-19 crisis continues and the country begins to reopen.
Yesterday CMS released a second interim final rule with comment period announcing a new round of regulatory waivers and rule changes to provide additional flexibility to the health care system as the COVID-19 crisis continues and the country begins to reopen. MACPAC has written to CMS administrator Seema Verma to express its concern that
MACPAC has written to CMS administrator Seema Verma to express its concern that  Earlier this month the state launched a $450 million Hospital Emergency Loan Program to provide short-term loans to hospitals struggling financially with their response to the COVID-19 emergency. Today the
Earlier this month the state launched a $450 million Hospital Emergency Loan Program to provide short-term loans to hospitals struggling financially with their response to the COVID-19 emergency. Today the  Quality Assurance and Performance Improvement (QAPI). (New since 4/21 Release) CMS is modifying certain requirements in 42 CFR §483.75, which requires long-term care facilities to develop, implement, evaluate, and maintain an effective, comprehensive, data-driven QAPI program. Specifically, CMS is modifying §483.75(b)–(d) and (e)(3) to the extent necessary to narrow the scope of the QAPI program to focus on adverse events and infection control. This will help ensure facilities focus on aspects of care delivery most closely associated with COVID-19 during the PHE.
Quality Assurance and Performance Improvement (QAPI). (New since 4/21 Release) CMS is modifying certain requirements in 42 CFR §483.75, which requires long-term care facilities to develop, implement, evaluate, and maintain an effective, comprehensive, data-driven QAPI program. Specifically, CMS is modifying §483.75(b)–(d) and (e)(3) to the extent necessary to narrow the scope of the QAPI program to focus on adverse events and infection control. This will help ensure facilities focus on aspects of care delivery most closely associated with COVID-19 during the PHE. Training and Assessment of Aides: (New since 4/21 Release) CMS is waiving the requirement at 42 CFR §418.76(h)(2) for Hospice and 42 CFR §484.80(h)(1)(iii) for HHAs, which require a registered nurse, or in the case of an HHA a registered nurse or other appropriate skilled professional (physical therapist/occupational therapist, speech language pathologist) to make an annual onsite supervisory visit (direct observation) for each aide that provides services on behalf of the agency. In accordance with section 1135(b)(5) of the Act, we are postponing completion of these visits. All postponed onsite assessments must be completed by these professionals no later than 60 days after the expiration of the PHE.
Training and Assessment of Aides: (New since 4/21 Release) CMS is waiving the requirement at 42 CFR §418.76(h)(2) for Hospice and 42 CFR §484.80(h)(1)(iii) for HHAs, which require a registered nurse, or in the case of an HHA a registered nurse or other appropriate skilled professional (physical therapist/occupational therapist, speech language pathologist) to make an annual onsite supervisory visit (direct observation) for each aide that provides services on behalf of the agency. In accordance with section 1135(b)(5) of the Act, we are postponing completion of these visits. All postponed onsite assessments must be completed by these professionals no later than 60 days after the expiration of the PHE. Pennsylvania Senate Democrats
Pennsylvania Senate Democrats  The FDA has posted
The FDA has posted  The Trump administration has released two new documents to support its plan for reopening the country and its economy:
The Trump administration has released two new documents to support its plan for reopening the country and its economy:  The Department of Health has issued
The Department of Health has issued  The Pennsylvania Health Law Center has launched a “
The Pennsylvania Health Law Center has launched a “ The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has shared its
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has shared its  On Wednesday night Governor Wolf addressed the state about his plan to gradually reopen Pennsylvania and its economy. While the governor had previously announced some reopenings, he outlined general parameters for authorizing additional steps and further business activity.
On Wednesday night Governor Wolf addressed the state about his plan to gradually reopen Pennsylvania and its economy. While the governor had previously announced some reopenings, he outlined general parameters for authorizing additional steps and further business activity. The CDC has published information about
The CDC has published information about